Affinity maturation services
Take an existing binder from micromolar to nanomolar or nanomolar to picomolar. Targeted mutagenesis libraries map every beneficial mutation and display selection confirms the best combinations.
Optimize your binder →Systematic protein binding optimization
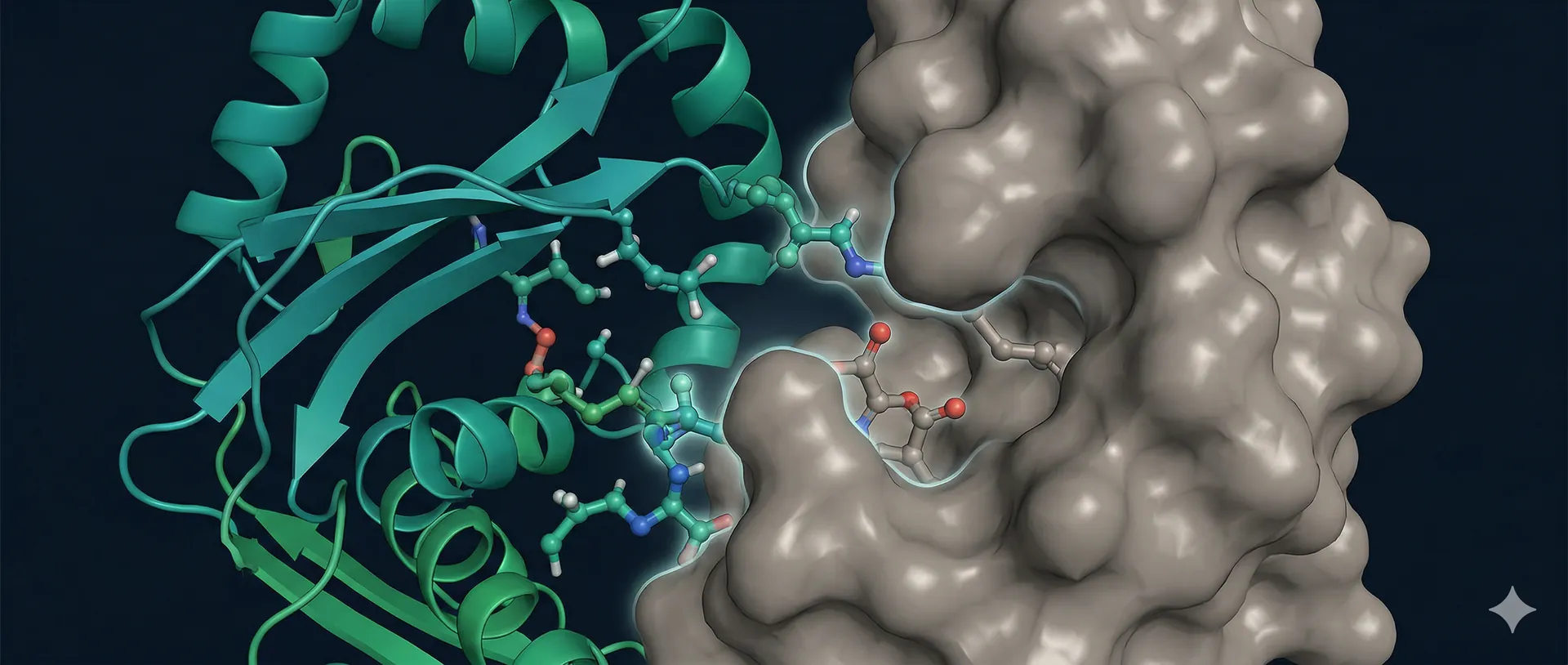
Traditional affinity maturation relies on random mutagenesis and iterative selection — a process that explores sequence space blindly and often requires many rounds to achieve meaningful improvements. Our approach replaces randomness with data.
A targeted mutagenesis library of your lead binder — saturation scanning, combinatorial CDR diversification, or computationally guided variants — is screened on yeast display under binding selection. The resulting fitness landscape reveals every single-point mutation that improves affinity at every position. The top beneficial mutations are then combined into a focused combinatorial library and screened again.
This two-stage approach — comprehensive single-mutant mapping followed by rational combination — consistently achieves 10-100 fold improvements in Kd while preserving specificity and expression.
Single-mutant scanning
Saturation or targeted mutagenesis library screened for binding. Identifies every affinity-improving substitution across the binding interface and framework.
Beneficial mutation ranking
Mutations ranked by enrichment score. Positions with multiple improving substitutions flagged for combinatorial exploration.
Combinatorial library
Top 5-15 beneficial positions combined in a focused library. Defined sequence panel, not random — every variant is a designed combination.
Stringent selection
Combinatorial library screened under limiting antigen concentrations. The most improved variants are those that maintain binding under competitive conditions.
Protein formats for affinity maturation
Nanobodies and VHH
Single-domain antibodies are ideal for maturation due to their small size (full saturation coverage in one library) and robust yeast display expression. Complete CDR and framework scanning in a single experiment.
scFv fragments
Single-chain variable fragments matured by scanning VH and VL CDR regions. Saturation or combinatorial scanning identifies improvements in both chains simultaneously, capturing inter-chain epistatic effects that single-chain optimization misses.
De novo designed binders
Computationally designed binders from RFdiffusion, BindCraft, or Boltzgen often bind in the low micromolar range. Iterative maturation through targeted mutagenesis and display selection is the standard follow-up to improve initial designs to application-relevant affinities.
Fab and IgG (via mammalian display)
Full antibody format maturation on mammalian display when native glycosylation or Fc function is important. Lower library diversity compensated by the biological relevance of hits selected in mammalian context.
Alternative scaffolds
DARPins, affibodies, fibronectin domains, and other non-immunoglobulin scaffolds. Our maturation approach is format-agnostic — any protein that can be displayed and sorted can be systematically optimized.
Bispecific constructs
Maturation of one arm of a bispecific while maintaining the other arm unchanged. Mutagenesis scanning focused on the target-binding interface of the arm being optimized.
Iterative display-based selection rounds
Each round of selection applies increasing stringency — lower antigen concentration, shorter incubation, or competitive off-rate selection — to progressively enrich for higher-affinity variants.
Concentration-based stringency
Sequential rounds at decreasing antigen concentrations. At limiting concentrations, only variants with improved on-rate or reduced off-rate maintain detectable binding. Multi-concentration titration series within a single sort provide apparent Kd rankings across the entire library.
Off-rate selection
Competition with excess unlabeled antigen after initial binding. Variants with slow off-rates retain labeled antigen longer and sort higher. Directly selects for the kinetic parameter most relevant to in vivo residence time and therapeutic efficacy.
A worked example: pH-dependent affinity maturation
Engineering pH-dependent binding via yeast surface display
640-clone targeted mutagenesis library, six FACS sort cycles, and convergent hotspot residues. Demonstrates the enrichment-score methodology and cumulative ranking that underpin our maturation campaigns.
Have a binder that needs tighter binding?
Share your lead sequence and current affinity data. We will design an affinity maturation campaign to reach your target Kd.
Start a project →